|
9/9/2023 0 Comments Graphical analysis physics lab 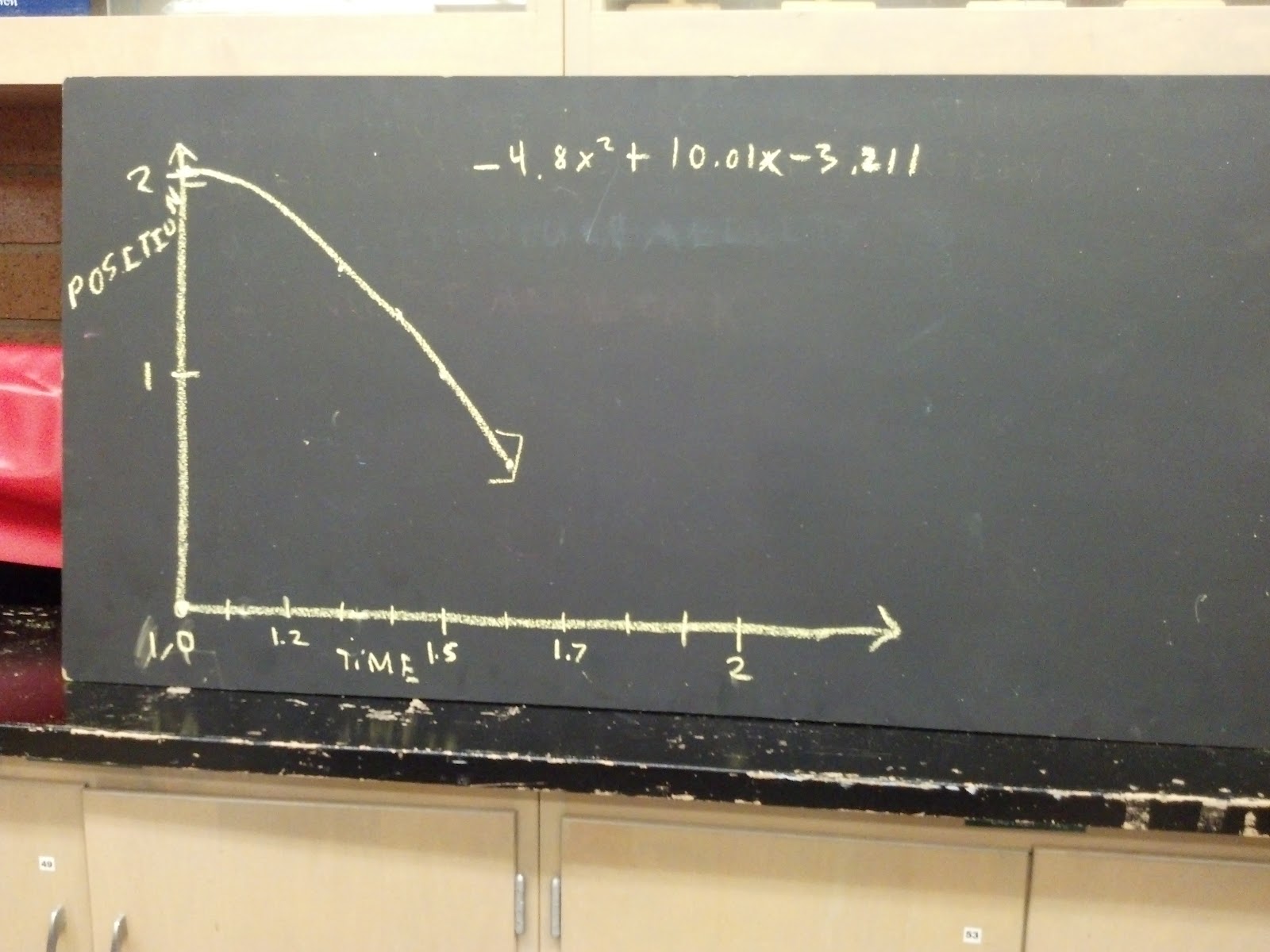
To decide what number to enter, look at your graph to see how far back along the x-axis you need to go in order to cover the area where volume = 10.5 L. In the section labeled Forecast enter a number in the box labeled Backward, since we want to extend the trendline the backward x direction.Click on the Layout tab along the top menu, then Trendline > More Trendline Options.Method (1): Extrapolate the trendline and estimate where the point on the line is. How can you find the temperature if it doesn't fall between the known points? There are two ways to do this. Note that the value 10.5 L falls outside the range of the plotted data. You are asked to determine the gas temperature. 
It can now be used to make certain predictions.įor example, suppose the 1 mole sample of helium gas is cooled until its volume is measured to be 10.5 L. The graph contains a visual representation of the relationship (the plot) and a mathematical expression of the relationship (the equation).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
